Wang, K., Zhan, Y., Huang, R. J.*, Wang, M., Ni, H. Y., Kampf, C. J., Cheng, Y. F., Bilde, M., Glasius, M., Hoffmann, T.: Molecular characterization and source identification of atmospheric particulate organosulfates using ultrahigh resolution mass spectrometry, Environ. Sci. Technol., 53, 6192-6202, 2019.
Recently, organosulfates (OSs) have been observed as substantial constituents of atmospheric organic aerosol (OA) in a wide range of environments. Due to the presence of a deprotonated functional group R-O-SO3−, OSs are acidic and highly water soluble and therefore may affect the surface activity and hygroscopic properties of the atmospheric particles, leading to potential impacts on climate. However, the chemical composition, sources and formation mechanism of OSs are still not well investigated, hindering a better understanding of the impacts of OSs on climate.
The chemical composition and sources of OSs in the ambient atmosphere is very complex caused by a wide variety of natural and anthropogenic sources as well as the complex multiphase chemical reactions. Hence, characterizing the chemical composition and source identification of OSs is a challenging but vital task.
In this study, Dr. Wang Kai and Prof. Huang Rujin created an ‘OS precursor map’ (Fig. 1) based on the elemental composition of previous OS chamber experiments, which can be used to identify the sources of OSs observed in the ambient aerosol samples. Then, according to this ‘OS precursor map’, they estimated the possible sources and molecular structures of OSs in atmospheric PM2.5 (particles with aerodynamic diameter ≤ 2.5 mm) samples, which were collected in urban areas of Beijing (China) and Mainz (Germany) and analyzed by ultrahigh performance liquid chromatography (UHPLC) coupled with Orbitrap mass spectrometer. Based on the ‘OS precursor map’, together with the polarity information provided by UHPLC, OSs in Mainz samples are suggested to be mainly derived from isoprene/glyoxal or other unknown small polar organic compounds, while OSs in Beijing samples were generated from both isoprene/glyoxal and anthropogenic sources (e.g., long-chain alkanes and aromatics). The nitrooxy-OSs in the clean aerosol samples were mainly derived from monoterpenes, while much fewer monoterpene-derived nitrooxy-OSs were obtained in the polluted aerosol samples, showing that nitrooxy-OS formation is affected by different precursors in clean and polluted air conditions.
This study provides a new insight to identify the possible precursors of OSs by creating this ‘OS precursor map’, which can help us to better understand the sources and formation of OSs. Meanwhile, additional information of elemental composition of OSs and nitrooxy-OSs identified in future chamber simulations can be used to further improve this ‘OS precursor map’.
This work was published in Environmental Science & Technology with link https://pubs.acs.org/doi/10.1021/acs.est.9b02628
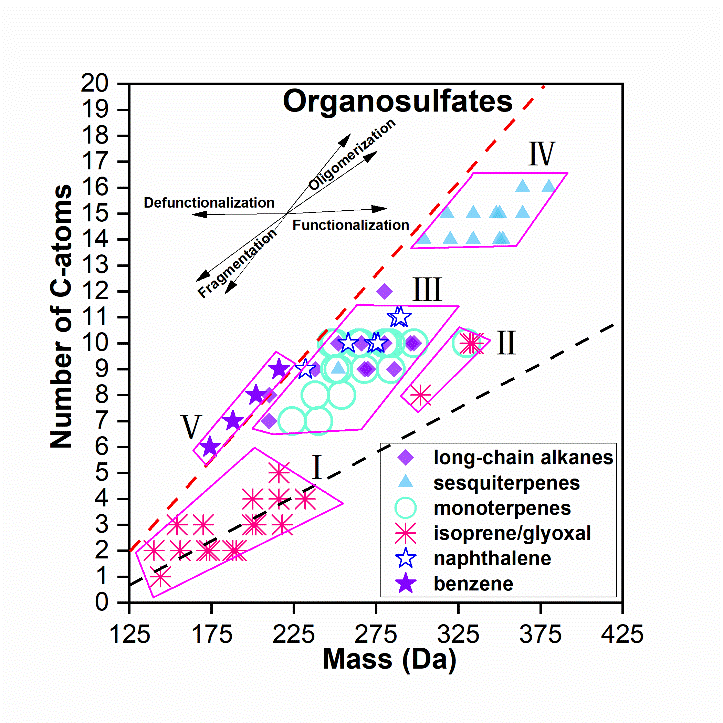
Figure 1. The OS precursor map represented by the molar mass and carbon number in the OS formulas.

